 
So now that we know how to do that, let's continue onward with calculating Mueller, Mass.). So these are the steps you must always use always employ in order to determine the molar mass of any compound you come face to face with.  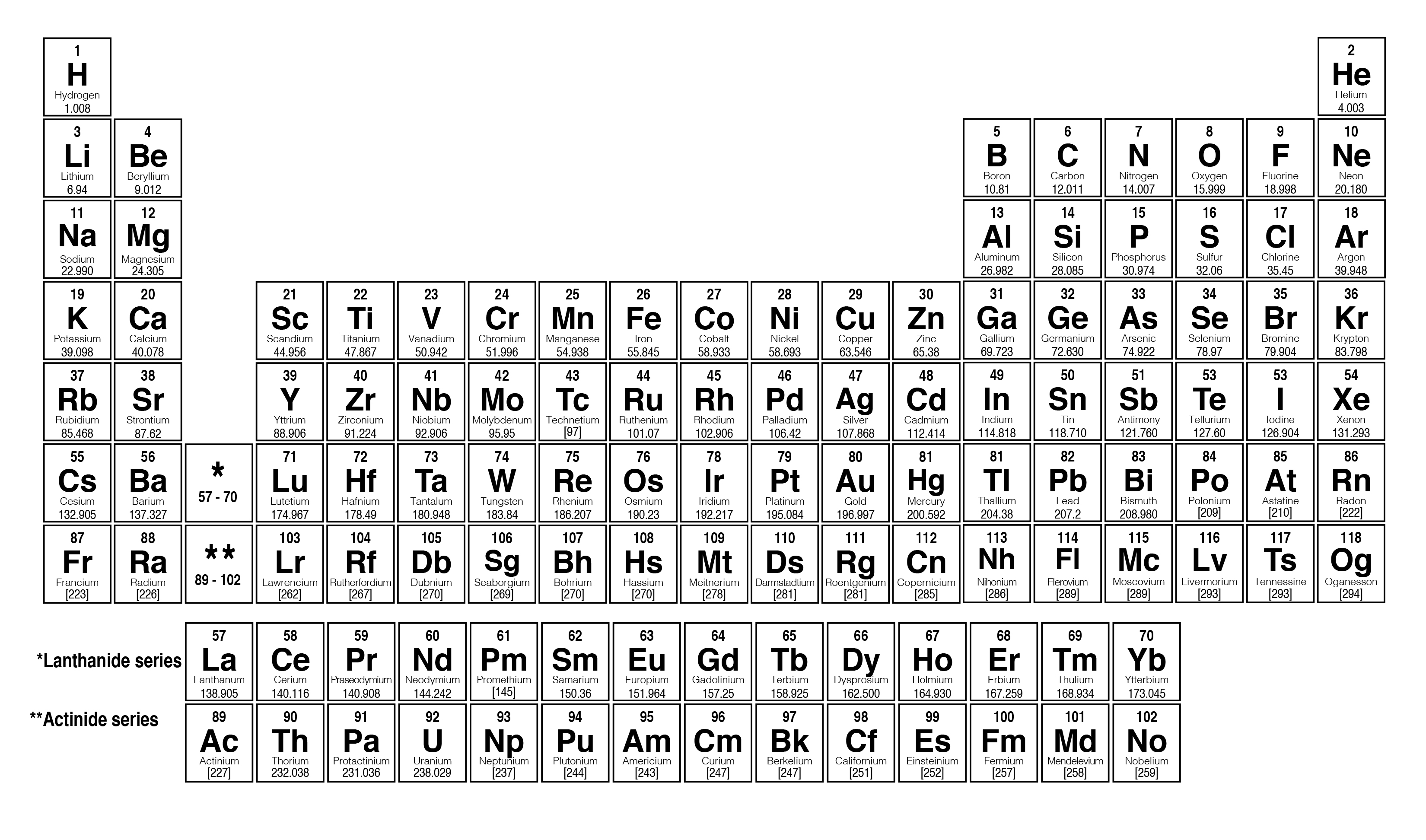
g per mole, so this would represent the Mueller Mass of our compound. When we do that, we're going to get a total of 1 32. Periodic Table with Mass Numbers and Atomic Symbols The table has mass, or molar mass, shown as the number in the bottom of each square. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2H5Cl: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214. So we take all these numbers, we add them all up together. Now that we have each of those totals, Step four is you add up the totals after multiplication to determine the molar mass off the compound. Show correct conversion factor you will use: B) Now show the calculation steps by using the name of the compound: 3. Finding molar mass starts with units of grams per mole (g/mol). Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. This site explains how to find molar mass. Formula Mass: Molar Mass: 2 Calculate the number of moles produced from 0.832 g of Na2SO4. The reason is that the molar mass of the substance affects the conversion. Now multiplying them with their atomic masses gives us totals here. Use Mass of an element up to two decimals from your periodic table. Answer - moles Check A sample of 0. g/mol Check Convert the value 46.2 grams NH, Clinto moles. Eight hydrogen, one sulfur and four oxygen's. Using a Periodic Table with Two Decimal Places, calculate the Molar Mass of Sr (CIO), with correct Significant Figures. So from step one, we found out we had to nitrogen. The numbers on top are the atomic numbers, so let's not worry about those next, you're gonna multiply together the number of each element with their atomic masses from the periodic table. So we have 14.1 g per mole, 1.8 g per mole, 32.7 g per mole and 16 roughly grams per mole for oxygen. I also usually use the full number given on the periodic table just to be exact as well, but I have noticed some people round their masses from the periodic table to about 2 or 3 decimal places. That's because it's the average of all the isotopes for that particular element. A whole number, usually has decimal places. Remember that the atomic mass is the number that is not. 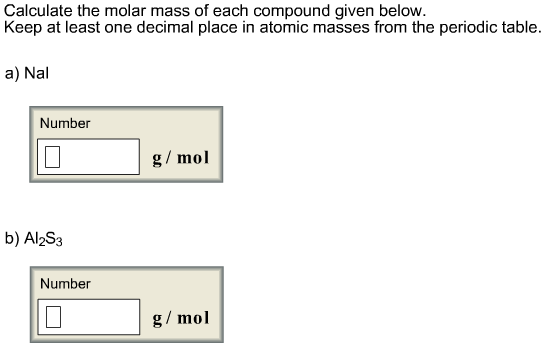
So we have nitrogen, hydrogen, sulfur and oxygen. Next, find the atomic masses of each element from the periodic table. Use the molar mass formula to calculate the mass: mass molar mass × number of moles. Use uppercase for the first character in the element and lowercase for the second character. Note that some masses are in parentheses, usually towards the bottom of the table of elements, The parentheses basically mean that scientists don’t. Generally, this number is the one with the decimal point. The table has mass, or molar mass, shown as the number in the bottom of each square. It will calculate the total mass along with the elemental composition and mass of each element in the compound. Periodic Table with Mass Numbers and Atomic Symbols. Enter the molecular formula of the substance. Since chlorine has a molar mass of 35.453 g/mol on the periodic table, the molar mass of the chlorine gas Cl 2 is twice this value. This program determines the molecular mass of a substance. So there we know that there's a one that we don't see, So there's just one sulfur and then we see that there's four oxygen's, so we've successfully counted each of these elements within the compound. To obtain this value, follow these steps: Determine the molar mass of the gas. Next, Let's look at the S 04 So s 04 there's only one sulfur there. We're done with everything in parentheses. So it becomes two times one which will give us to nitrogen and then we have to times four, which will give us eight hydrogen. That means we have one nitrogen, four hydrogen and this sub script of to what happens here is that too gets distributed. Alright, So within these parentheses we have NH four. Now, if the elements are within parentheses, just remember to distribute the subscript thio each not to each element. Alright, So step one, you have to count the number of each element within the given compound. Question says, calculate the molar mass of the compound NH 42 s 04 In order to do that, we're gonna follow each of the following steps. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
